Writer: Ian C. Langtree – Author/Editor for Disabled World (DW)
Revealed: 2026/04/20
Publication Sort: Informative
Class Matter: Weightloss – Associated Publications
Contents: Synopsis – Introduction – Predominant – Insights, Updates
Synopsis: In lower than 5 years, the pharmacological therapy of weight problems has gone from a quiet nook of inner drugs to essentially the most disruptive class in American well being care, and the merchandise arriving in 2026 are rewriting what clinicians, courts, and insurers can credibly say about weight, potential, and the equipment of the human physique – Disabled World (DW).
- Matter Definition: Weight Loss Treatment
-
A weight reduction treatment is a prescription or over-the-counter pharmaceutical product authorised by a nationwide drug regulator, such because the U.S. Meals and Drug Administration, to assist adults, and in some circumstances adolescents, scale back extra physique weight and keep that discount over time when mixed with a reduced-calorie weight loss program and elevated bodily exercise. These drugs work by means of a variety of mechanisms, together with mimicking intestine hormones that regulate urge for food and insulin response, suppressing urge for food by means of central nervous system pathways, or blocking the intestinal absorption of dietary fats, and they’re delivered as weekly or every day injections, oral tablets, or capsules relying on the precise product.
Introduction
Weight Loss Medicine 2026: Manufacturers, Outcomes, Incapacity
Weight problems care in the US has been rewritten in lower than a decade. The pharmacological therapy of weight problems used to imply phentermine prescriptions and over-the-counter orlistat on the drugstore, with modest outcomes that usually vanished the second a affected person stopped taking the capsule. That world nonetheless exists, nevertheless it now sits alongside a era of injectable and oral drugs that routinely produce double-digit share weight reduction and are reshaping how physicians, insurers, and courts take into consideration physique weight itself (Weight problems Drugs Affiliation, 2026).
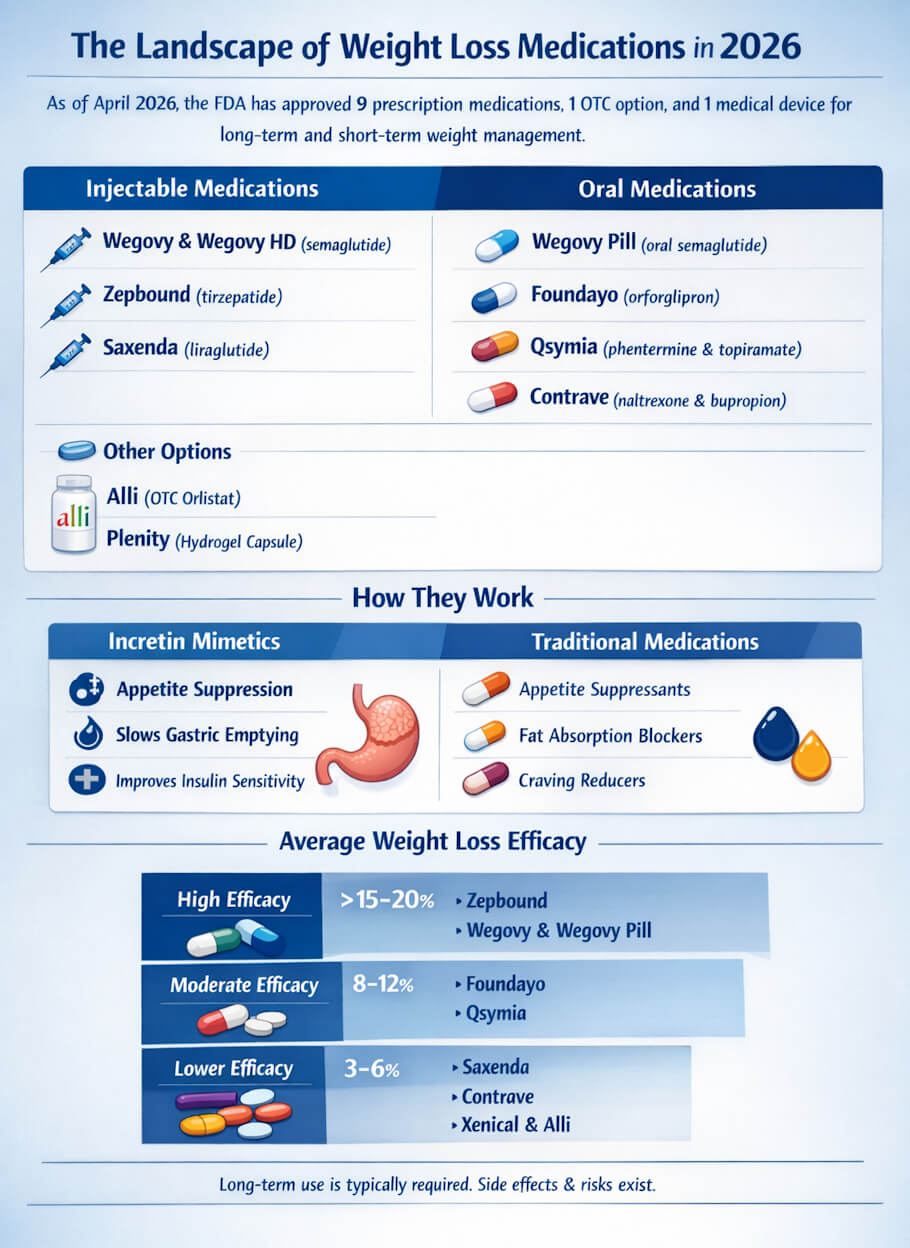
As of April 2026, the U.S. Meals and Drug Administration has authorised 9 pharmacological choices for long-term or short-term weight administration in adults, together with one medical machine categorised as a prescription product and a single over-the-counter possibility. Two of these drugs arrived inside the final 5 months: the oral formulation of Wegovy in December 2025 and Foundayo (orforglipron) on April 1, 2026 (U.S. Meals and Drug Administration, 2026a). The next-dose injectable model of Wegovy, branded Wegovy HD, was authorised on March 19, 2026 (U.S. Meals and Drug Administration, 2026b). The pipeline behind these merchandise is crowded. Analysts at Ozmosi have recognized roughly three dozen GLP-1-related weight administration candidates in growth, with new launches anticipated yearly for the foreseeable future.
Predominant Content material
How These Drugs Really Work
Many of the newer weight reduction medication belong to a category referred to as incretin mimetics. They imitate hormones the intestine releases after meals – glucagon-like peptide-1 (GLP-1) and, in some circumstances, glucose-dependent insulinotropic polypeptide (GIP). These hormones gradual gastric emptying, blunt urge for food alerts within the mind, and enhance insulin sensitivity. The older drugs work in a different way: some are stimulants that suppress urge for food (phentermine), some block fats absorption within the gut (orlistat), and a few mix neuromodulators that scale back reward-driven consuming (Contrave).
The sensible distinction that issues most to sufferers is less complicated than the pharmacology. A few of these merchandise are injected beneath the pores and skin, normally as soon as every week. Others are swallowed. Some require a prescription and doctor supervision. Just one true anti-obesity drug is bought with no prescription in the US.
Injectable Prescription Drugs
Wegovy (semaglutide)
Manufactured by Novo Nordisk and authorised in 2021 for continual weight administration, Wegovy delivers semaglutide as soon as weekly by subcutaneous injection utilizing a pre-filled pen. Part 3 trials reported imply weight discount of roughly 14.9% of physique weight over 68 weeks on the 2.4 mg dose. The drug is authorised for adults with a physique mass index (BMI) of 30 or greater, or 27 or greater with at the least one weight-related comorbid situation, and for adolescents 12 and older with a BMI at or above the ninety fifth percentile. In March 2026 the FDA authorised the next 7.2 mg dose referred to as Wegovy HD, which produced further weight discount past the usual dose in scientific testing (U.S. Meals and Drug Administration, 2026b).
Zepbound (tirzepatide)
Eli Lilly’s Zepbound, authorised for weight problems in late 2023, is at present the simplest weight reduction treatment on the U.S. market. It’s a twin GLP-1 and GIP receptor agonist administered as soon as weekly by injection. Within the head-to-head SURMOUNT-5 trial revealed in The New England Journal of Drugs, tirzepatide produced a imply weight discount of 20.2% at 72 weeks in contrast with 13.7% for semaglutide, the primary direct scientific comparability of the 2 (Aronne et al., 2025). A separate section 3 trial in members with knee osteoarthritis and weight problems discovered weight losses of as much as 28.7% with the associated triple-agonist retatrutide, which stays in growth however alerts the place this class is heading.
Saxenda (liraglutide)
Liraglutide, marketed by Novo Nordisk as Saxenda, was the primary GLP-1 receptor agonist authorised for weight administration in adults and can be authorised for adolescents 12 and older. Not like Wegovy and Zepbound, it should be injected every day quite than weekly. Its weight reduction impact is extra modest, sometimes 5% to eight% of physique weight at 56 weeks, nevertheless it stays related for sufferers who can not tolerate the newer brokers or whose insurance coverage favors it on formulary.
Oral Prescription Drugs
Wegovy Tablet (oral semaglutide for weight problems)
In December 2025 the FDA authorised an oral formulation of Wegovy, the primary oral GLP-1 receptor agonist cleared particularly for continual weight administration. On the highest dose, trial members misplaced a mean of 16.6% of their physique weight after 64 weeks, and roughly one in three misplaced at the least 20% (AARP, 2026). The capsule is taken as soon as every day on an empty abdomen with a small quantity of water, a protocol some sufferers discover inconvenient. A lower-dose oral semaglutide product referred to as Rybelsus has been obtainable since 2019 for sort 2 diabetes and is usually prescribed off label.
Foundayo (orforglipron)
Authorised on April 1, 2026, Foundayo is Eli Lilly’s oral GLP-1 receptor partial agonist and the one weight reduction capsule of its class that may be taken any time of day with out meals or water restrictions. Its chemistry – a small-molecule nonpeptide quite than a peptide – makes it extra secure within the abdomen. Within the ATTAIN-1 trial, adults taking the best dose of 36 mg misplaced a mean of 12.4% of physique weight, about 27 kilos, after 72 weeks, in contrast with about 2 kilos for placebo (Eli Lilly and Firm, 2026). Foundayo was the primary new molecular entity authorised beneath the FDA’s Commissioner’s Nationwide Precedence Voucher program, which shortened the evaluation timeline by a number of months (U.S. Meals and Drug Administration, 2026a).
Qsymia (phentermine and topiramate)
Qsymia combines a controlled-substance urge for food suppressant (phentermine) with the anticonvulsant topiramate in an extended-release capsule. It’s authorised for continual weight administration in adults and in pediatric sufferers 12 and older. Imply weight reduction in trials runs about 8% to 10% at one yr. It requires prescriber enrollment in a Threat Analysis and Mitigation Technique due to the teratogenic potential of topiramate.
Contrave (naltrexone and bupropion)
Contrave pairs a low-dose opioid antagonist with an antidepressant to scale back urge for food and cravings. It produces roughly 5% to six% imply weight reduction at one yr. The mix can increase blood strain and is contraindicated in sufferers taking opioids, so clinician choice issues.
Phentermine (Adipex-P, Lomaira)
Phentermine is the oldest and least expensive prescription weight reduction treatment nonetheless in extensive use, initially authorised in 1959. It’s a sympathomimetic amine bought as Adipex-P, Lomaira, and beneath its generic title. It’s FDA authorised just for short-term use, usually as much as 12 weeks, due to its stimulant profile and potential for tolerance. Many clinicians prescribe it in cycles or alongside topiramate. It stays standard partly as a result of generic month-to-month prices can fall beneath $15 with out insurance coverage.
Xenical (prescription orlistat)
Orlistat, marketed on the 120 mg prescription power as Xenical, inhibits pancreatic lipase and blocks absorption of about 30% of dietary fats. Common weight reduction is 3% to five% at one yr. Its gastrointestinal unwanted side effects – oily stools, flatulence, fecal urgency when sufferers eat high-fat meals – are well-known and are the explanation many sufferers discontinue it.
Over-the-Counter and System-Based mostly Choices
Alli (orlistat 60 mg)
Alli is the one over-the-counter weight reduction treatment authorised by the FDA. It accommodates orlistat at half the prescription power and is obtainable on drugstore cabinets for adults 18 and older with a BMI of 25 or greater. Its efficacy and aspect impact profile mirror these of Xenical at a smaller magnitude. Alli stays helpful for sufferers who desire a nonprescription possibility and are prepared to take care of a low-fat weight loss program throughout therapy.
Plenity (prescription, not an OTC drug)
Plenity is commonly talked about alongside drugs however is technically a prescription medical machine quite than a drug. It’s a hydrogel manufactured from carboxymethylcellulose and citric acid that expands within the abdomen to create a sense of fullness. Sufferers take three capsules with water earlier than lunch and dinner. Imply weight reduction within the gateway trial was about 6.4%. As a result of it’s categorised as a tool, it’s regulated and reimbursed in a different way than the GLP-1 merchandise.
How the Drugs Evaluate for Efficacy
Ranked roughly by common p.c physique weight reduction at one yr or longer, the present choices fall into three tiers. On the prime sit Zepbound at about 20% imply loss, adopted by injectable Wegovy at about 15% and the Wegovy capsule at about 17% on the highest dose (Aronne et al., 2025; AARP, 2026). Foundayo sits close to the center at 12.4%, a significant however smaller impact than the injectables (Eli Lilly and Firm, 2026). Qsymia, Saxenda, and Plenity land within the 5% to 10% vary. Contrave, Xenical, and Alli sometimes produce 3% to six%.
These numbers are averages. Particular person response varies extensively. Roughly one in 5 sufferers on GLP-1 medication is a low responder, and as much as a 3rd lose greater than 20% of their beginning weight. Weight regain after discontinuation is frequent and infrequently substantial, which is why most clinicians body these drugs as continual therapies for a continual illness quite than brief programs.
The Overlap Between Weight Loss and Incapacity
Weight enters incapacity legislation by means of a number of doorways, and none of them are extensive. The Individuals with Disabilities Act (ADA) doesn’t record weight problems as a protected situation, and federal courts have been reluctant to deal with physique weight alone as a qualifying incapacity.
Weight problems Below the Individuals with Disabilities Act
The ADA defines incapacity as a bodily or psychological impairment that considerably limits a number of main life actions. The implementing laws of the Equal Employment Alternative Fee particularly exclude bodily traits inside a traditional vary, together with weight, except they end result from a physiological dysfunction. 4 federal circuit courts – the Second, Sixth, Seventh, and Eighth – have held that weight problems shouldn’t be an ADA impairment except it’s brought on by an underlying physiological situation (Richardson v. Chicago Transit Authority, 2019). Essentially the most cited case, Richardson, concerned a Chicago bus driver whose weight rose above 550 kilos; the courtroom declined to acknowledge his weight problems as a incapacity as a result of he produced no proof of a physiological trigger.
State and municipal legislation fills among the hole. New York Metropolis banned weight-based employment discrimination outright in 2023. Michigan and a handful of cities together with San Francisco, Washington D.C., and Binghamton keep related protections. Courts in Texas, Louisiana, Mississippi, California, New Jersey, New York state, Washington, and Oregon have been extra prepared than their federal counterparts to acknowledge weight problems, or perceived weight problems, as a qualifying situation in incapacity discrimination claims (Ogletree, 2023).
Weight-Associated Comorbidities That Do Qualify
The place weight itself falls outdoors the ADA, the circumstances that generally accompany it usually don’t. Sort 2 diabetes, obstructive sleep apnea, extreme osteoarthritis of the knees or hips, congestive coronary heart failure, lymphedema, and continual kidney illness can every independently meet the ADA definition of incapacity once they considerably restrict a serious life exercise. A warehouse employee with knee osteoarthritis extreme sufficient to restrict standing or strolling is protected beneath the statute whatever the quantity on the dimensions. This distinction is legally necessary and, in observe, is what number of massive workers pursue lodging quite than arguing weight problems by itself phrases.
How Weight Loss Treatment Can Change the Incapacity Image
The brand new era of GLP-1 drugs is beginning to alter the scientific calculus round weight-related incapacity. Past weight discount, semaglutide has demonstrated improved cardiovascular outcomes and diminished development of diabetic kidney illness, and tirzepatide has been proven to scale back liver fats, enhance metabolic dysfunction-associated steatohepatitis, and reduce the severity of obstructive sleep apnea (American Diabetes Affiliation, 2024). The TRIUMPH-4 trial of retatrutide reported substantial discount in knee osteoarthritis ache alongside weight reduction. As these indications increase, extra sufferers might discover that the underlying impairments limiting their work lives – joint ache, apnea-driven fatigue, uncontrolled diabetes – reply to a weekly injection. That doesn’t make the drugs an alternative choice to affordable lodging. It does imply the scientific image of a disabled employee with weight problems is much less static than it was 5 years in the past.
Entry, Insurance coverage, and the Sensible Obstacles
Protection is the quiet disaster beneath the thrill about these medication. Record costs for Wegovy and Zepbound run roughly $1,000 to $1,350 monthly earlier than rebates. Medicare Half D traditionally excluded weight reduction medication by statute, although that has begun to shift for obesity-related circumstances corresponding to heart problems. Business protection is uneven. Producer self-pay packages have narrowed the hole: Foundayo launched at $149 monthly for its lowest dose on self-pay and $25 monthly with the producer financial savings card for these with business insurance coverage (Eli Lilly and Firm, 2026). A FAIR Well being evaluation revealed in Might 2025 reported that greater than 2% of U.S. adults had stuffed a prescription for a GLP-1 weight reduction treatment within the previous yr, a share that has since grown.
For sufferers who qualify as disabled beneath state legislation or the ADA by means of an underlying situation, the calculus shifts once more. Some state Medicaid packages now cowl anti-obesity drugs when a comorbid incapacity is documented, and several other federal lodging claims have efficiently argued that denial of efficient weight administration therapy constitutes a failure to accommodate a physiological situation.
Dangers and Lifelike Expectations
None of those drugs is benign. Gastrointestinal unwanted side effects – nausea, vomiting, constipation, diarrhea – are the commonest cause sufferers discontinue GLP-1 medication. Rarer however severe dangers embody pancreatitis, gallbladder illness, and a boxed warning for thyroid C-cell tumors noticed in rodent research. Orlistat could cause liver harm in uncommon circumstances and interferes with absorption of fat-soluble nutritional vitamins. Phentermine carries the cardiovascular dangers typical of stimulants. Qsymia requires being pregnant prevention counseling resulting from topiramate. Contrave is contraindicated in sufferers with seizure issues or consuming issues.
Muscle loss throughout speedy weight discount is a priority particular to the GLP-1 class. Clinicians now routinely advocate resistance coaching and a minimal of 30 grams of protein per meal throughout energetic therapy (American Diabetes Affiliation, 2024). The mix of an amylin analog, cagrilintide, with semaglutide – referred to as CagriSema – is in late-stage growth and has reported imply weight lack of 20.4% at 68 weeks with much less muscle loss than semaglutide alone in early outcomes (Weight problems Drugs Affiliation, 2026).
Weight reduction treatment works greatest when sufferers, physicians, and payers deal with weight problems as what the American Medical Affiliation has referred to as it since 2013: a continual illness. That framing has not but absolutely reached the courts, and it has solely partly reached the insurance coverage market. However it’s the scientific actuality the following decade of those merchandise can be constructed on.
References:
AARP. (2026, April 2). FDA approves new weight reduction capsules: Prices and outcomes. AARP Well being.
American Diabetes Affiliation. (2024). Beginning FDA-approved weight administration drugs. Skilled Sources.
Aronne, L. J., Horn, D. B., le Roux, C. W., Ho, W., Falcon, B. L., Gomez Valderas, E., Das, S., Lee, C. J., Glass, L. C., Senyucel, C., and Dunn, J. P. (2025). Tirzepatide as in contrast with semaglutide for the therapy of weight problems. The New England Journal of Drugs, 393(16), 1543 – 1554.
Eli Lilly and Firm. (2026, April 1). FDA approves Lilly’s Foundayo (orforglipron), the one GLP-1 capsule for weight reduction that may be taken any time of day with out meals or water restrictions [Press release]. PR Newswire.
Ghusn, W., De La Rosa, A., Sacoto, D., Cifuentes, L., Campos, A., Feris, F., Hurtado, M. D., and Acosta, A. (2022). Weight reduction outcomes related to semaglutide therapy for sufferers with chubby or weight problems. JAMA Community Open, 5(9), e2231982.
Weight problems Drugs Affiliation. (2026, April 2). Prime weight reduction drugs. OMA Scientific Sources.
Ogletree Deakins. (2023). Weight problems as a incapacity beneath the Individuals with Disabilities Act. Employment Regulation Insights.
Richardson v. Chicago Transit Authority, 926 F.3d 881 (seventh Cir. 2019).
U.S. Equal Employment Alternative Fee. (2008). Enforcement steering on the Individuals with Disabilities Act and the ADA Amendments Act. Washington, DC: EEOC.
U.S. Meals and Drug Administration. (2026a, April 1). FDA approves first new molecular entity beneath Nationwide Precedence Voucher program [Press release].
U.S. Meals and Drug Administration. (2026b, March 19). FDA approves fourth product beneath Nationwide Precedence Voucher program, greater dose semaglutide [Press release].
College of Chicago Regulation Assessment. (2024). Weighing in: Why weight problems must be thought-about a qualifying incapacity beneath the Individuals with Disabilities Act. The College of Chicago Regulation Assessment, 91(3), 789 – 828.
Editorial Observe: None of those drugs is a completed reply to weight problems, and none resolves the uncomfortable questions on who will get entry, what insurance coverage can pay for, and the place the legislation attracts the road between a physique and a incapacity – however the subsequent a number of years will drive each a kind of questions into the open, whether or not the system is prepared or not – Disabled World (DW).
 Writer Credentials: Ian is the founder and Editor-in-Chief of Disabled World, a number one useful resource for information and knowledge on incapacity points. With a world perspective formed by years of journey and lived expertise, Ian is a dedicated proponent of the Social Mannequin of Incapacity-a transformative framework developed by disabled activists within the Nineteen Seventies that emphasizes dismantling societal boundaries quite than focusing solely on particular person impairments. His work displays a deep dedication to incapacity rights, accessibility, and social inclusion. To be taught extra about Ian’s background, experience, and accomplishments, go to his full biography.
Writer Credentials: Ian is the founder and Editor-in-Chief of Disabled World, a number one useful resource for information and knowledge on incapacity points. With a world perspective formed by years of journey and lived expertise, Ian is a dedicated proponent of the Social Mannequin of Incapacity-a transformative framework developed by disabled activists within the Nineteen Seventies that emphasizes dismantling societal boundaries quite than focusing solely on particular person impairments. His work displays a deep dedication to incapacity rights, accessibility, and social inclusion. To be taught extra about Ian’s background, experience, and accomplishments, go to his full biography.